Le palper-rouler est une nouvelle tendance pour perdre du poids dont tout le monde en parle. Mais qu’est-ce que c’est réellement ? Et comment ce massage s’effectue ? Dans cet article, on répond à toutes vos interrogations. D’abord, il faut savoir que le palper-rouler...
patientsorganizations.org


Santé
Beauté
Vous êtes sans cesse à la recherche de nouvelles trouvailles beauté ? Soyez la plus belle version de vous-même en découvrant nos adresses et conseils beauté !
Equilibre
Vous menez une vie à cent à l’heure et ne comptez pas vos heures ? Gardez l’équilibre et ne négligez pas votre santé !
Bien-Être
Un petit coup de mou ? Besoin de repos ? Laissez-vous guider par nos astuces bien-être pour être toujours au top !
Quels sont les tarifs du palper-rouler manuel ?
Le palper-rouler est une nouvelle tendance pour perdre du poids dont tout le monde en parle. Mais qu’est-ce que c’est réellement ? Et comment ce massage s’effectue ? Dans cet article, on répond à toutes vos interrogations. D’abord, il faut savoir que le palper-rouler...

Quels sont les tarifs du palper-rouler manuel ?
Le palper-rouler est une nouvelle tendance pour perdre du poids dont tout le monde en parle. Mais qu’est-ce que c’est réellement ? Et comment ce massage s’effectue ? Dans cet article, on répond à toutes vos interrogations. D’abord, il faut savoir que le palper-rouler...
Le coin Santé

Mon expérience avec l’hypnose opératoire : une alternative surprenante à l’anesthésie traditionnelle
Hypnose, anesthésie, conscience, suggestion, bras… Ces termes vous semblent peut-être étrangers, mais ensemble, ils tracent l’expérience dont nous allons vous parler aujourd’hui. Une expérience qui a changé notre regard sur la santé, et plus particulièrement sur la...
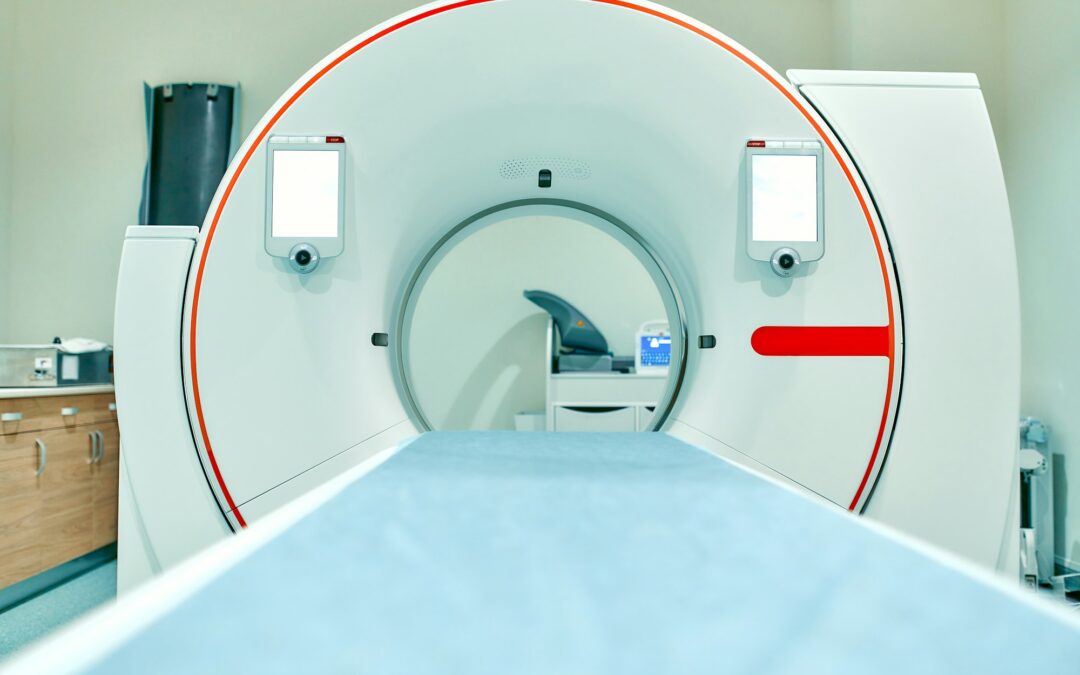
L’Intelligence Artificielle dans le domaine de la santé
L’intelligence artificielle (IA) et l’apprentissage automatique sont en train de révolutionner le domaine de la santé. Ils permettent d’améliorer les diagnostics, les traitements médicaux et la vie des patients. Cet article se propose de vous faire découvrir comment...
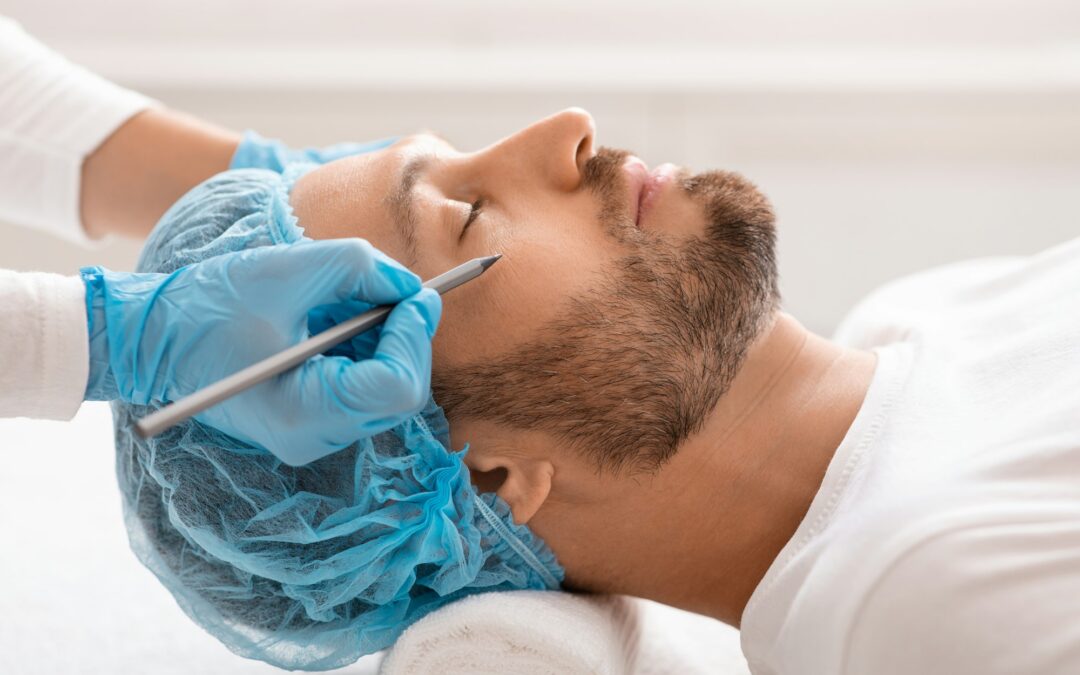
Qu’est-ce que la blépharoplastie : le remodelage du regard ?
La blépharoplastie, vous avez peut-être déjà entendu ce terme mais ne savez pas vraiment de quoi il s'agit… Cette chirurgie esthétique des yeux consiste à enlever l'excès de peau et/ou de graisse au niveau des paupières supérieures et/ou inférieures. Cette opération...
Le coin Beauté

Masque cheveux à la banane : Découvrez les différentes recettes et les bienfaits du masque capillaire
Pourquoi utiliser un masque à la banane pour vos cheveux? La banane, ce fruit délicieux est également un allié précieux pour vos cheveux. Mais pourquoi donc? Vous demandez-vous. Les bananes sont riches en vitamines et minéraux bénéfiques pour la santé de vos cheveux....

LPG résultats au bout de combien de séances : Combien de séances sont nécessaires ?
Aujourd'hui, nous allons nous pencher sur une question qui préoccupe nombre de personnes désireuses d'améliorer leur apparence physique : combien de séances sont nécessaires avant de voir des résultats visibles avec le traitement LPG pour le corps ou le visage ? C'est...

Opération du nez : quels sont les retours d’expérience ?
Aujourd'hui un très grand nombre d'hommes et de femmes qui ont subi une opération du nez, ou du moins qui s'y intéressent fortement. En effet, si vous consultez les avis partagés en ligne sur ce genre d'intervention esthétique, vous serez étonné du nombre de...
Le coin Bien-être
Poche gel bain dérivatif : Informations sur les poches en gel spécifiquement conçues pour les bains dérivatifs et où les trouver.
Pour débuter, une petite mise en situation. Vous êtes assis chez vous, une journée d'été, la température oscille autour des 30°C. Vous ressentez cette chaleur qui envahit votre corps, cette sensation inconfortable d'être en permanence en transpiration. Vous pensez à...

Thérapie par la musique : Se relier aux émotions et aux sensations grâce à la musique.
La musique a toujours été un moyen universel de communication et de connexion émotionnelle. Elle est capable de faire ressentir des émotions puissantes et d'apporter du réconfort lors des moments difficiles. La thérapie par la musique, ou musicotherapie, est une...

Rollfit résultats : Quels sont les résultats que l’on peut obtenir grâce à l’utilisation régulière du Rollfit
Le 04/09/2023, je vous propose un voyage à travers l'univers du Rollfit. Cet équipement de fitness qui a conquis de nombreux amateurs d'activités physiques. Mais quels sont réellement les résultats que l'on peut obtenir grâce à l'utilisation régulière du Rollfit et...
Partenaires
Nos Suggestions
Pour vos problèmes bricolage, n’hésitez pas à contacter notre partenaire alginate dentaire
Notre coup de cœur beauté/bien être
Les meilleurs astuces beauté et bien-être sur Beauté fatale : santé, vitalité, confiance en soi, rajeunissement, traitements.